8 Key Elements of an Effective QMS
Inside This Article
 Compliance issues can create significant barriers for medical device companies looking to bring new products to market. In 2020 alone, the U.S. Food and Drug Administration (FDA) issued 422 citations or observations (i.e., Form 483) to medical device companies, indicating that their products were in potential violation of regulatory requirements1. These FDA observations typically stem from deficiencies in a company’s quality management system (QMS) and cover areas such as corrective and preventive actions (CAPAs), complaint handling procedures, and product nonconformance procedures.
Compliance issues can create significant barriers for medical device companies looking to bring new products to market. In 2020 alone, the U.S. Food and Drug Administration (FDA) issued 422 citations or observations (i.e., Form 483) to medical device companies, indicating that their products were in potential violation of regulatory requirements1. These FDA observations typically stem from deficiencies in a company’s quality management system (QMS) and cover areas such as corrective and preventive actions (CAPAs), complaint handling procedures, and product nonconformance procedures.
Having an effective QMS in place is critical to help companies mitigate these compliance risks and achieve commercialization success. Whether your organization is setting out to implement a new QMS or fine-tune an existing one, you should keep these key elements in mind.
Quality Management Systems (QMS) are evolving with trends in sustainability, agile manufacturing, cloud-based solutions, advanced analytics, big data, and AI automation. These trends aim to enhance product quality, efficiency, and responsiveness to market changes, while emphasizing eco-friendly practices and real-time data integration.
QMS Essentials for Life Sciences Companies
1. Quality Policy and Objectives
When developing a quality policy, it should state your organization’s overall purpose and mission and support the company’s strategic direction. It should also state your commitment to continuous improvement as well as meeting all applicable regulatory and customer requirements. Ultimately, a quality policy serves as the framework for the entire QMS.
Quality objectives are measurable goals that stem directly from the quality policy. Quality objectives should contribute to the enhancement of customer satisfaction and are commonly tied to areas such as:
- Product nonconformities or defects
- Product performance
- Efficiency
- Safety
- Delivery
- Customer service
For each of these areas, establish quantifiable targets and link to key performance indicators (KPIs) that help monitor the achievement of the objectives over time. It is best practice to make quality objectives SMART: Specific, Measurable, Achievable, Relevant, and Time-bound.
Here is an example of a quality objective that is tied to customer service:
- Objective: Reduce customer complaints
- Target: Achieve 25% reduction within the next three months
- KPIs: Customer call reports or customer satisfaction surveys
The setting of objectives and targets involves individuals from key functional areas across the organization. Once established, quality objectives should be clearly documented and communicated throughout your organization. It is also important to update quality objectives as your business needs continue to evolve.
2. Quality Manual
A quality manual documents the scope of the QMS. The manual should include information about your organization’s objectives, expectations, policies, processes, and more. The manual should also include requirements needed for your organization to comply with ISO, FDA, and other regulatory standards.
3. Organizational Structure and Responsibilities
A QMS should outline the roles and responsibilities of the key stakeholders, resources, and infrastructure that are needed to achieve your quality objectives. It should also provide a clear visual of how your organization is structured. This can be in the form of a flow chart or similar type of diagram.
4. Internal Processes
Identify and define processes that require any type of resource to transform inputs into outputs. Resources can encompass employees, machines, or technology. Establish and document standards and metrics for measuring the performance of these processes to ensure that quality outputs are always the end result.
5. Customer Satisfaction With Product Quality
As part of QMS, organizations are required to monitor customer satisfaction to determine if their quality objectives are being met. This can be in the form of customer satisfaction surveys, customer complaint reports, customer review websites, and other monitoring systems.
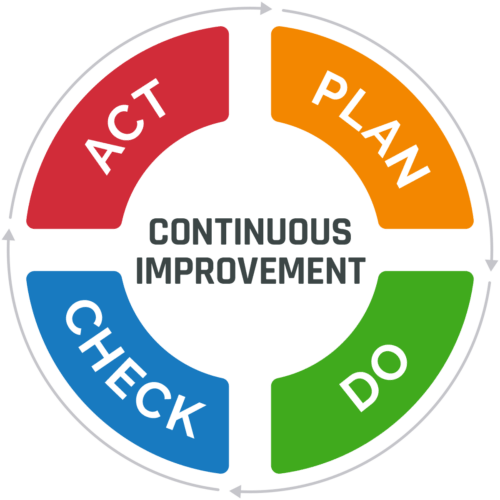
6. Continuous Improvement
Continuous improvement is the ongoing improvement of products, services, or processes through incremental changes over time or changes that occur all at once. A widely used tool for continuous improvement is a four-step quality assurance method called the plan-do-check-act (PDCA) cycle:
- Plan: Identify an opportunity for improvement and plan for change
- Do: Implement the change on a small scale
- Check: Use data to analyze the results of the change
- Act: If the change was successful, implement it on a wider scale and continuously assess the results. If the change does not work, begin the cycle again.
Other common tools include Six Sigma, lean, and total quality management. These approaches emphasize employee engagement and teamwork and practices to reduce variation, defects, waste, and cycle time.
7. Document Control
Have a proper system in place for document control. This includes:
- Review and approval of documents for adequacy prior to release
- Review/update/reapproval of documents on an as-needed basis
- Identification of changes and current document revision status
- Preventing obsolete documents from unintended use
- Policy or procedure for the identification, storage, protection, retrieval, retention, and disposal of documents
8. Enterprise Quality Management System (eQMS) Software
Once you have established your quality policy and objectives and other key processes for your quality management system, how do you ensure that it yields the desired results?
Today, more and more businesses are relying on some type of eQMS software to centralize all of the key elements of their quality and product records and allow for more effective collaboration across dispersed teams and partners.
Cloud-based enterprise platforms such as Arena QMS help eliminate nonconformance issues, reduce audit risks, and improve new product development processes by improving quality controls and documenting policies, procedures, and practices.
To gather additional insights on implementing an effective QMS, read this free eBook and read our QMS common questions.
References
