How Cloud QMS Simplifies Validation
Why the need to validate?
In the journey to deliver safe and effective devices to market, medical device manufacturers must simultaneously manage a myriad of regulations. To maintain quality compliance throughout the manufacturing process, quality leaders have the option to use various types of quality management systems (QMS). They can leverage manual, paper-based ones; on-premises software; or cloud-based QMS software. The FDA doesn’t regulate what type of system manufacturers use, just that they have a quality system that ensures products consistently meet applicable requirements and specifications.
For software-based solutions, however, the FDA does require medical device manufacturers to prove, or validate, that the quality system meets the requirements for its intended use. Depending on the type of QMS solution, the validation process can pose added burdens on teams, draining resources, and incurring added costs. When evaluating new software systems, many manufacturers do not consider the cost and impact of validating software. However, overlooking the impact of validating software can mean the difference between product introduction success or failure. How do today’s medical device innovators comply with FDA regulations while also meeting burdensome validation requirements? A cloud-based QMS software designed to be validated easily, even with regular enhancements, is the best option to deliver high-quality, compliant products to market.
On-Premises QMS software increases burdens on quality teams
When a medical device company purchases a cutting-edge QMS software solution, they expect to take advantage of capabilities that improve quality and increase transparency and historical tracking of information as they develop and release new devices. However, they too often forget to analyze the options to get new software enhancements or upgrades and to evaluate the initial impact and ongoing cost of validating the software.
With advanced analytics and reporting features, quality management software for manufacturing enables companies to make informed decisions based on real-time quality data. Organizations can track key performance indicators, monitor supplier performance, and identify trends to optimize production processes. This data-driven approach improves efficiency, reduces risks, and fosters continuous improvement across the manufacturing lifecycle.
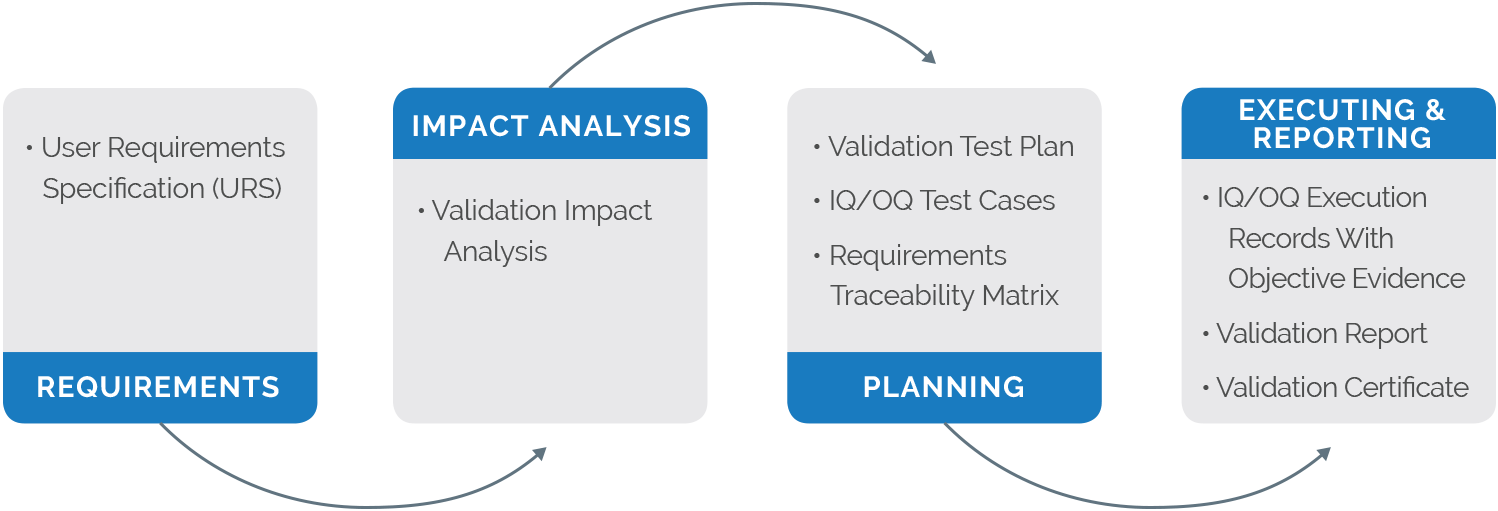
The FDA mandates that software used in the production or implementation of a device must be validated. This applies to the initial implementation of the software as well as future upgrades. Faced with many years of horror stories upgrading on-premises software systems, device manufacturers often rely on outdated paradigms to base future validation decisions. So, quality teams are often hesitant to upgrade their QMS software because of the associated burdens of validation.
Traditional validation of on-premises solutions for each and every new enhancement of software can lead to more than a drain on current resources. For some companies, the time, expense, and effort associated with validating new versions outweighs the desire to take advantage of new functionality, even when that functionality will streamline quality and product-release processes. As a result, too many companies continue to rely on an outdated QMS software system that fails to keep up with technological advances, making it harder for the manufacturer to stay ahead of the competition.
Why cloud multi-tenant QMS simplifies validation
As cloud-based applications become more sophisticated, secure, and affordable, quality leaders should evaluate the advantages of using a multi-tenant solution that adheres to current best practices in software validation, security, and innovation. Cloud-based QMS software with multi-tenant architecture provides the best way to take advantage of ongoing advancements in technology while providing the lowest total cost of ownership (TCO) with a simplified validation experience. With a multi-tenant cloud solution, medical device companies can leverage software enhancements regularly without the traditional validation “heavy lifting” required by older on-premises or single-tenant QMS solutions.
Arena’s cloud-based QMS is a multi-tenant solution, designed to provide medical device companies with regular enhancements to continually improve business processes. With a level of shared infrastructure for all QMS customers, the validation process is made easier by leveraging common test protocols for both installation qualification (IQ) and operational qualification (OQ) requirements.
When Arena QMS is paired with Arena’s software validation offering, Arena Validate, customers benefit from a better validation experience that reduces manual effort and time-consuming work. Removing traditional validation barriers enables companies to keep pace with the latest technological and functional features and frees quality and regulatory resources to focus on innovation, market responsiveness, and continual improvement to deliver high-quality products.
Conclusion
LNS Research found that software validation is one of the top challenges faced when trying to speed product development from early research and development to commercialization. When quality leaders don’t have to worry about validating software, they can focus on core competencies and, ultimately, accelerating the delivery of innovative and safe devices to market.
Still curious about the specific ways Cloud QMS can simplify software validation? Check out our FDA Software Validation: How Cloud QMS Reduces Costs and Resource Drain white paper to learn more.
Sources