In addition to managing effective projects, many companies are faced with everincreasing regulatory compliance requirements. Failure to successfully oversee regulatory compliance issues can not only delay or derail product launches, it can result in warnings, fines, and even company shutdowns. For example, the widely publicized problems at Theranos can be attributed to the fundamental inability of the company’s products to comply with FDA regulations. Even minor regulatory issues can create serious delays getting products to market. PRG’s Mike Keer explains one example where a company recently designed a product it thought would have no difficulty passing UL and FCC emissions. However, the product failed the emissions tests three times. Each failure required a redesign of several weeks, including engineering time and building a new prototype. The issues were not major, but they still set the product launch back three months, which significantly reduced the revenues generated by the product.
Ensuring your product meets applicable regulations is often the responsibility of quality assurance and regulatory assurance teams with expertise in monitoring and complying with constantly evolving regulations and standards. These teams drive the validation process to guarantee that products and development processes meet regulatory requirements. Building a compliance-first culture across the product lifecycle ensures that regulatory alignment, team collaboration, and proactive risk management are embedded from design through delivery.
Arena helps simplify and expedite both regulatory (e.g., FDA, ISO) and environmental compliance (e.g., RoHS, REACH, WEEE, Conflict Minerals) by managing the required product, training, compliance, and quality records in a single system.
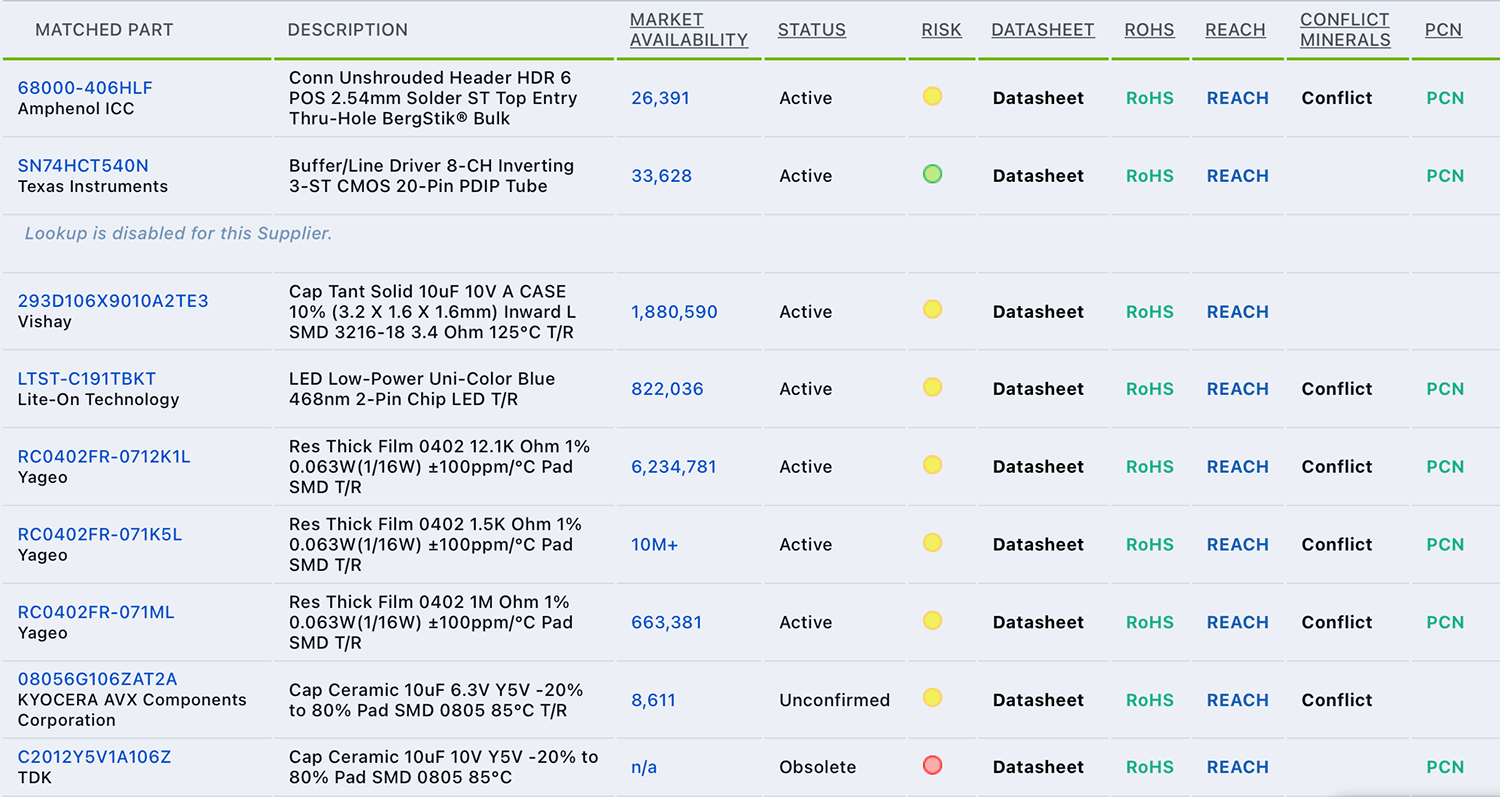
Arena’s Supplier Item Lookup provides regulatory and material availability information to eliminate risks to ensure compliant parts can be sourced when needed.
Arena also electronically tracks and maintains requirements, test details, and electronic approvals linked to the appropriate part of the product record. Arena tracks design control elements and deliverables for the impacted product, quality, and production teams.
This simplifies regulatory compliance and ensures responsible team members are always aware of key action items and project deliverables. Maintaining regulatory information in context with the product record makes it easy to respond to and pass audits, ultimately speeding product launches. Furthermore, for medical device manufacturers, Arena manages device master records (DMRs), design history files (DHFs), and validation maintenance services to simplify FDA compliance and software validation. This ensures quality processes meet intended uses per FDA Title 21 CFR Part 820 (i).