SOLUTIONBRIEF
Competing in today’s global economy requires greater connectivity between your teams and systems. Getting high quality products to market, while adhering to industry standards and regulatory agencies, is critical to your success. Arena Quality is designed to control, support, and maintain your quality management processes. With Arena, these quality processes are connected directly to the product record and development process. Connecting product processes with corrective action preventive action (CAPA) processes accelerates resolution of quality issues and drives continuous improvement throughout the product lifecycle.
Key benefits |
What Arena Quality Does |
|
| Drives Continuous Quality Improvement |
|
|
| Improves Quality Process Visibility |
|
|
| Accelerates Issue Resolution |
|
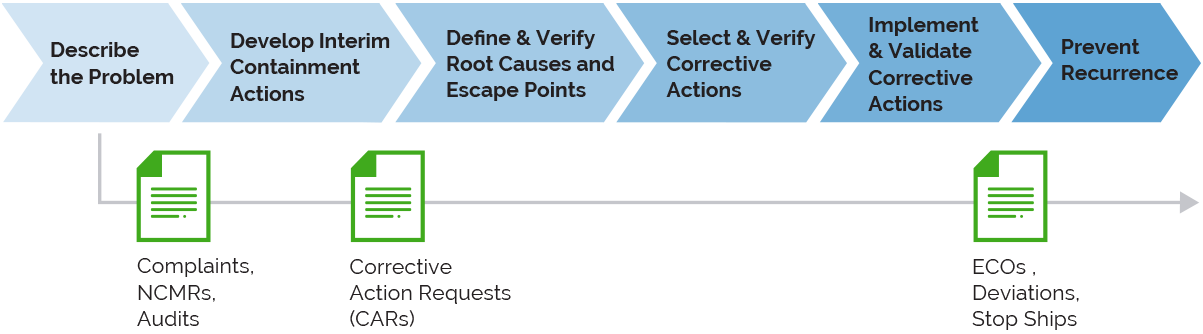
Arena’s connected quality management makes it easy to identify, prevent, and resolve quality issues using a single source of quality and product truth. Your internal teams and partners can act on quality information faster with full traceability to drive continuous improvement.
With Arena Quality, you can keep your primary focus on developing and delivering safe and effective products to your customers. Don’t wait any longer. Start using Arena Quality today.
To find out how you can join our community of innovative customers, contact [email protected] or call 1.866.937.1438.
Connects continuous improvement into your business processes with product development and quality processes managed in a single system. Directly link your quality processes to your product development and sustaining efforts.
Drive the complete corrective action process to resolution with issue identification, root cause analysis, due dates, and explicit sign-off, including 21 CFR Part 11 compliant electronic approvals.
Manage quality processes across all products to keep quality issues under control, while solving problems across shared parts and assemblies. Control supplier access to individual quality processes and share selectively for maximum security. Easily monitor and track quality issues for suppliers and the items they source.
Collaborate on quality with your entire team to unite engineering, operations, quality, and other groups. Your entire team can see quality issues, actions, and processes in-context with the complete product record. Impacted teams can view critical information about the affected parts or products — including changes, assemblies, quality actions, SOPs, requirements, defects, and suppliers—from any quality issue, action, or change record.
Configure industry-standard or customizable quality approval processes with company-specific steps and terminology. Assign responsibility at multiple levels.
Maintain a complete history of quality issues, non-conformances, and corrective actions.