3 Reasons to Integrate eQMS, PLM, and RIM Platforms

As you’re beginning to read this post, you likely have many applications running on your computer that help manage your day-to-day tasks. Whether it’s to chat with colleagues, manage files, or host a video conference, it’s undeniable that the rapid growth of technology has revolutionized the way we work.
In a recent report, Okta Inc. found that the number of software applications used across all companies grew 22% over the past four years, with large enterprises now having an average of 175 different applications. As a result, the question of “how can software applications help me do my job?” has evolved to a more important, and often more complex, question of “how can my software applications work together to support the work that I need to do?” Your software applications can no longer work in isolation, given the network of information that now exists within an organization.
Application Overload Impacts Regulatory Affairs Productivity
For regulatory affairs professionals, this question has never been more important to address. A recent Deloitte study found that on average, 30-50% of a regulatory professional’s time is spent on administrative and non-value-added activities, and most of this time was spent simply looking for information inside of the organization. This number is understandable when you think about the large amount of data that a regulatory professional is responsible for consolidating, and the rapidly growing number of information sources within the organization.
As technology stacks continue to grow, regulatory affairs teams must develop an integration and workflow automation strategy to maintain productivity. In this post, we’ll outline three key benefits that come from integrating the most used tools by regulatory affairs teams—electronic quality management (eQMS), product lifecycle management (PLM), and regulatory information management (RIM). If you’ve missed initial posts on the benefits of these tools, check out 5 Signs You’re Ready to Take Your Regulatory Team Digital and Top 6 Benefits of a RIM System.
3 Reasons to Integrate Your Regulatory Software Stack Today
Reason #1: Reduce time to market and improve revenue
When working to register and sell a product within a new market, the goal is to do so quickly and efficiently. This is easier said than done, given the amount of ever-changing information that needs to be compiled to support a regulatory submission. With new products and evidence files being created and updated daily, you’re working on an application as the data is moving from beneath you. One way to ensure you’re working efficiently on new product registrations is to create harmony between your systems by synchronizing product master data from your PLM to your RIM platform. With these systems connected, you avoid worrying about manually adding new SKUs to your RIM platform to include within your market application, once it’s available in your PLM.
This synchronization automatically pulls in the relevant SKUs, with the associated information that allows you to add it to a working application, including renewals and amendments. As a regulatory professional, understanding that you’re working with the full set of applicable products lets you manage your regulatory data at the SKU level.
This allows you to have a trusted, single source of truth for all the regulatory data surrounding your products, including relevant UDI requirements, associated standards, and applicable essential principles. The result is a newfound state of nirvana for managing compliance: Your new market applications match the products your company wants to sell, with no wasted time searching for this information.
Reason #2: Better compliance with global regulations
A common reason that products don’t get registered and launched quickly is simply due to the changing regulatory landscape. As we all know, the initial application and approval is only the beginning of a product’s regulatory lifecycle. Once a product launches, the responsibility then shifts to maintaining compliance, even as those requirements are updated. On top of this, you have information that is constantly updated within your organization as it relates to the product, specifically the technical documentation and evidence that was used to get it approved in the first place.
This is where integrating your eQMS and RIM systems provides significant relief. Rather than waiting on a note from your quality or engineering teams, when integrated, your RIM platform can notify you directly when eQMS’ quality records change, allowing you to quickly run reports on every associated submission and conduct an impact assessment. Rather than spending valuable time identifying where your company may be affected, your time is spent conducting the impact assessment itself.
Integrating your eQMS doesn’t just help you understand the impact of changing documentation, it helps you throughout your regulatory processes. Once integrated, you can synchronize your existing technical evidence to your products and working market applications to avoid version control-related errors and manual uploads. Whether you’re working on a 510k, STED, ToC, or any other market-related application, the evidence you need to include is just a click away, allowing you to link the file from your eQMS directly within your RIM platform.
With this integration set up you no longer need to manually sift through tens of thousands of files to find the one you need. Instead, you can easily search by title and file number to find what you’re looking for in seconds. What results is a system that helps manage and support your workflows—not interrupt them. You’ll be able to respond quickly to regulatory changes, ensure that products can stay in-market, and maintain MDSAP compliance.
Reason #3: Increase cross-company collaboration
Regulatory affairs professionals are responsible for understanding relevant regulatory requirements and how their company will fulfill them. This means you inevitably need to work closely with your colleagues in other teams to help support regulatory activities. While you can reach out to your quality and engineering teams to ask questions and ensure that you’re working with the latest information and correct file versions, this communication takes time. When you add in busy schedules and conflicting time zones, waiting for answers can become another roadblock in your path to finalizing a new product submission.
The integration of your PLM, eQMS, and RIM systems ensures that your regulatory, quality, and engineering teams are all working from the same product set and technical file versions. You transition from relying on one-off email or chat conversations to an agreed upon, shared set of information that is consistent across teams. Finally, when all functional groups are working from the same tools with the same set of information, your organization can more easily align standard operating procedures to manage all product-related regulatory information.
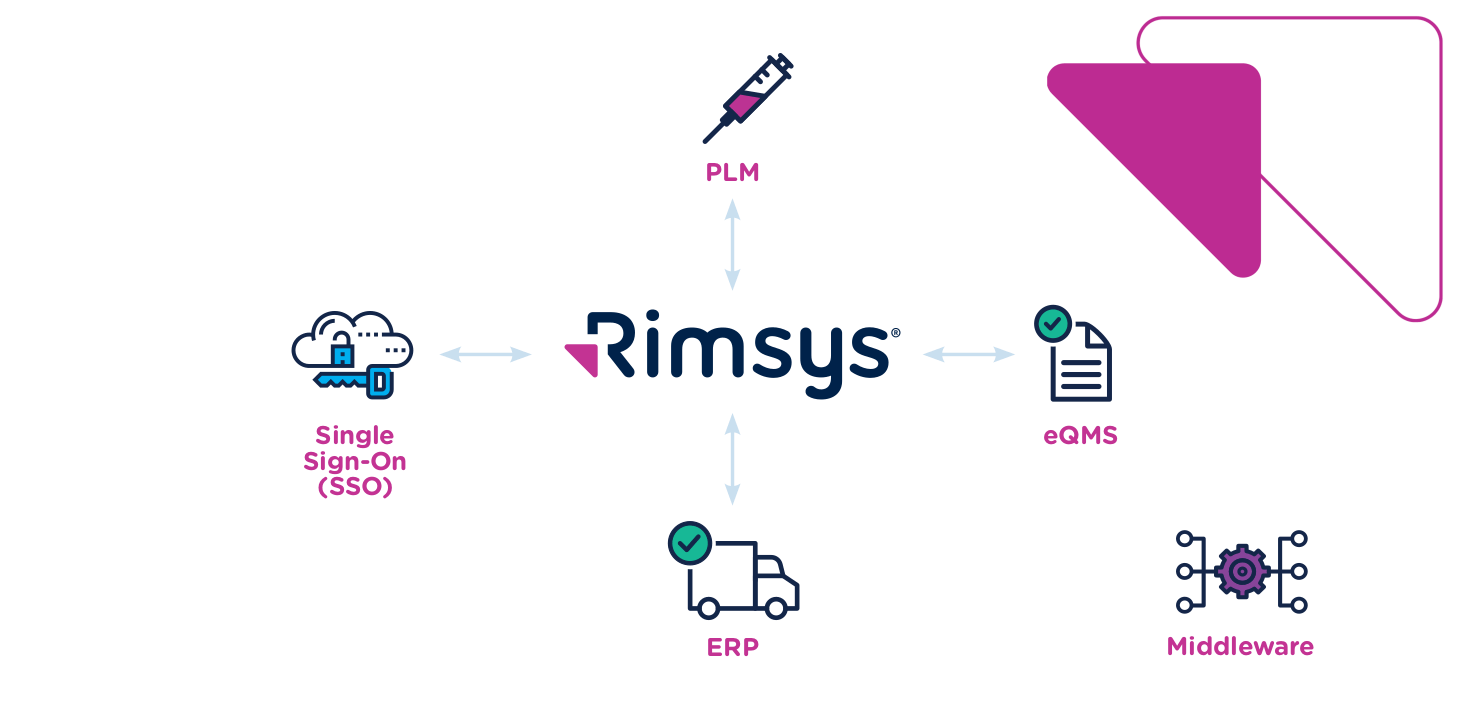
Integration: Valuable and Essential for Regulatory Affairs Teams
As you can see, there are compelling benefits associated with integrating the primary tools in your regulatory affairs stack: PLM, eQMS, and RIM. You can speed time to market, improve regulatory compliance, lower risk, and drive more effective communication and collaboration across teams. But the reality is that most teams can’t afford not to invest in these integrations. Establishing and maintaining regulatory compliance has always been a challenge, but as product lifecycles shorten, companies look to move more quickly, and critical information gets spread across an ever-growing number of systems, it will become impossible without better automation and integration.
The good news is that many modern systems are built with integration in mind. RIM software from Rimsys, for example, provides native connectivity across a wide range of systems, including Arena PLM and eQMS solutions. To see how you can benefit from better integrations, request a custom demo today and check out the ultimate guide to PLM software.
