No More Guesswork: How Arena’s BOM History Delivers Full Product Traceability
Inside This Article

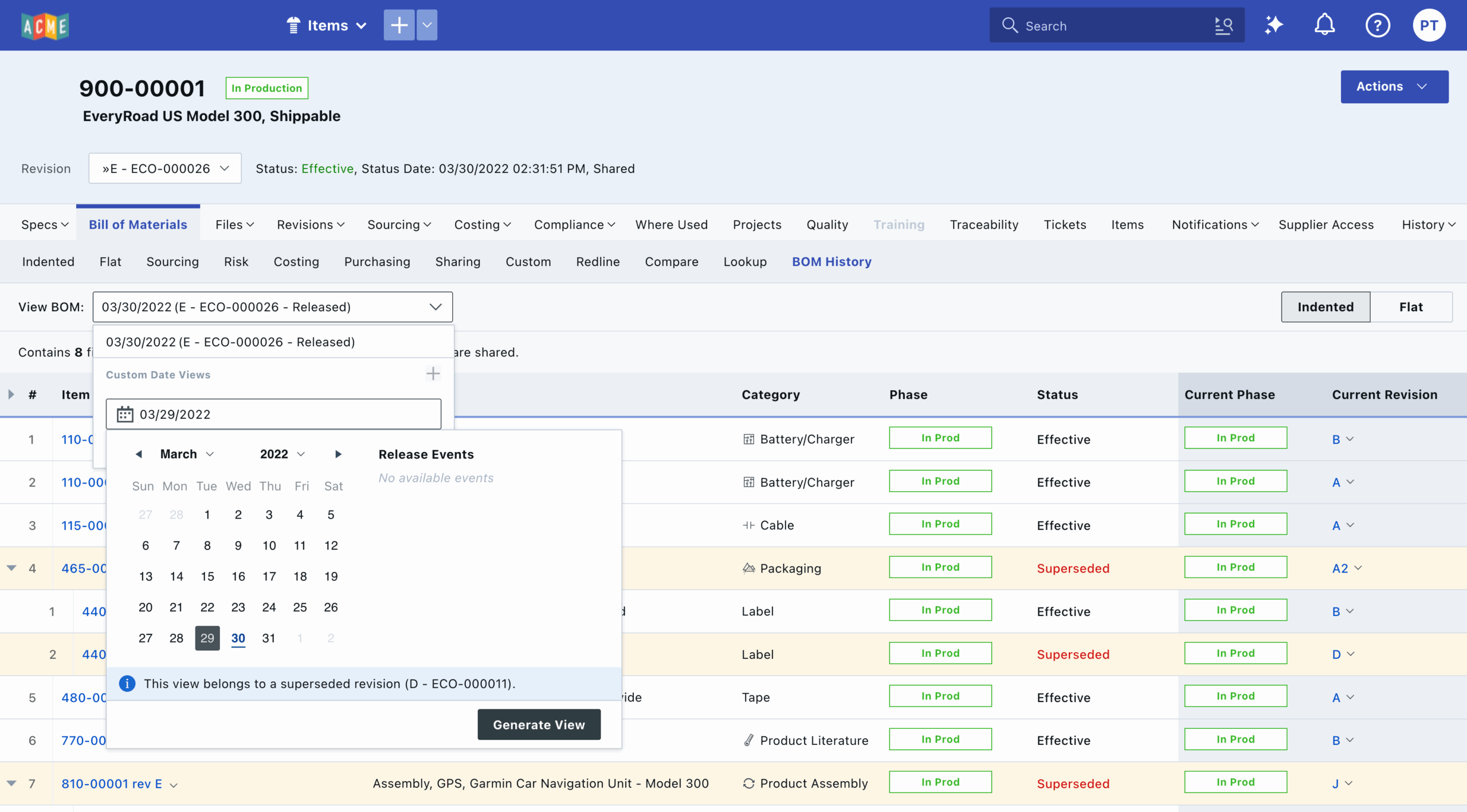
Ever tried to trace a product’s exact build from two years ago—only to find there’s no easy way since most components reference the latest effective revision? That’s the reality many product teams navigate, and it can affect everything from regulatory audits to field service operations. The bill of materials (BOM) history capability in Arena’s latest product lifecycle management (PLM) and quality management system (QMS) software release addresses this issue. With a clear, time-stamped record of every product configuration, you’ll know exactly what was built, when, and with which part revision.
We caught up with Senior Product Manager Pär Svensson to unpack how this new capability transforms the way companies manage their products throughout the lifecycle.
What inspired the development of the BOM history feature in Arena PLM?
Pär: The introduction of BOM history arose from our customers’ desire to know the exact product configuration and effective part revisions that were manufactured at a specific point in time. Typically, PLM users would view past BOMs, but they always referenced the latest component and subassembly revisions. By accessing historical BOM data within Arena, customers can streamline their procurement, field service, and compliance processes even further.
How does BOM history improve procurement for product companies?
Pär: With BOM history, companies can access a time-stamped record of every BOM revision, including the exact components and subassemblies used in each manufactured unit. This transparency enables procurement teams to make more informed purchasing decisions. It also helps prevent costly mistakes by ensuring that everyone is working from the same, accurate data set. Arena’s centralized product record means that sourcing teams are no longer hampered by siloed or disconnected spreadsheets.
In what ways does BOM history support field service operations?
Pär: Field service teams often need to know exactly what component revisions are inside the product to diagnose issues, perform repairs, or conduct preventive maintenance. With BOM history, they gain instant access to the precise configuration of any product as it was manufactured, speeding up troubleshooting and ensuring that the right parts are available for service calls. This improved visibility translates directly into better customer support and faster resolution times.
How does BOM history enhance warranty analysis?
Pär: Warranty analysis depends on knowing what was delivered to the customer. If a product fails, it’s crucial to trace back to the specific BOM revision used in that unit. BOM history allows companies to quickly identify which components were present, analyze failure rates, and determine whether issues are isolated or systemic. By having a complete historical record, companies can proactively address potential issues and reduce warranty costs over time.
What impact does BOM history have on compliance reviews?
Pär: Compliance is a major concern, especially for companies in regulated industries like medical devices, aerospace and defense, and high-tech electronics. BOM history simplifies compliance by providing a clear, auditable trail of each product configuration and ensuring every change is tracked and documented. This supports corrective and preventive action (CAPA) processes, internal audits, and external regulatory reviews. It’s all about demonstrating accountability and transparency which builds trust with industry regulators.
How does BOM history contribute to broader efforts like improving customer lifetime value, strengthening supply chain resilience, or supporting M&A activities?
Pär: Having a clear record of historical BOM data means companies can deliver better support, faster service, and more reliable products—all of which help keep customers happy. BOM history also improves supply chain resilience by making it easier to track component updates, source compliant parts, and respond quickly to disruptions.
In the context of mergers and acquisitions, BOM history provides the documentation needed to demonstrate process maturity and maximize business value. Essentially, having documented, easily accessible historical data makes the process smoother.
What feedback have you received from customers about BOM history?
Pär: Customers have expressed appreciation for the improved visibility and traceability that Arena’s BOM history provides. They’ve noted that it speeds up investigations, audits, and service calls while reducing the risk of errors and miscommunication. The ability to access historical BOM data in real time has been described as a game-changer for teams across engineering, procurement, quality, and field service.
What are the next steps for companies looking to use BOM history in Arena PLM?
Pär: Companies should start by evaluating their current BOM management processes and identifying areas where historical data could improve decision-making. By leveraging BOM history, companies can unlock new efficiencies, strengthen compliance, and deliver greater value to their customers.
Closing Thoughts
As Pär highlighted, the ability to access and analyze historical BOM data empowers teams to make better sourcing decisions, expedite compliance reviews, and strengthen customer relationships. For companies ready to take their product lifecycle management to the next level, Arena’s BOM history is a powerful feature for driving success.
