What truly makes Arena unique is the ability to manage linked relationships between DMRs, DHFs, BOMs, individual components, AML/AVL, documentation, product history, and any changes or quality issues. Anyone with appropriate access privileges can view the related quality, change, and product records to understand better the actions required to resolve problems. Leveraging information quickly and easily facilitates robust change control, supply chain collaboration, quality management, and key product-related business processes. Consider how Arena QMS addresses the following business needs and external requirements.
Engineering creates and documents product designs comprising the complete product record, and then everyone else uses this information to verify, validate, source, manufacture, and support the products. But who is everyone else? They can be teams from manufacturing, quality, regulatory, clinical, purchasing, supply chain partners, and field service groups.
When evaluating solutions, consider every aspect of the product. The complete product record includes BOMs that define electro-mechanical assemblies with reference designators to identify a component in an electrical schematic or on a printed circuit board (PCB). The product record is further enhanced with the supporting manufacturers’ information (i.e., relational AML/AVL), drawings, specifications, and procedures.
With Arena, every team has instant access to the complete product record, which means they can perform their jobs more quickly and more accurately. When an issue arises, Arena empowers impacted teams to resolve the issue. For example, if there is a sourced component quality issue, purchasing can easily view the AML to find and select approved alternate parts.
As companies grow, they use different systems to manage different elements of the product record. The BOMs may be in spreadsheets on a controlled, shared drive. The AML may be in ERP with changes managed by email correspondence. And the engineering changes may be logged in yet another system. Quality processes, like CAPA, often require a complete view of the product.
With Arena, product companies can eliminate the use of multiple disparate systems. Arena provides a single, scalable enterprise platform to manage the items, BOMs, DMRs, DHFs, and all associated product documentation. This simplifies control, supports quality processes, and increases traceability throughout the product lifecycle.
Ensuring your internal teams and partners are trained and can prove competency is required for FDA 21 CFR Part 820. Recent FDA audits reveal that more findings relate to training processes than to personnel training. In 2019, the FDA reported 64 training-related findings3. Inadequate “procedures for training and identifying training needs” resulted in 37 findings and a lack of “personnel training documentation” was cited 24 times.
Arena Training manages training records so managers know which employees have been trained on applicable standard operating procedures (SOPs), policies, and manufacturing process instructions. Arena provides a single, comprehensive solution to manage training processes and records to ensure you are always in compliance, can respond to audits easily, and guarantee that your employees are continually trained.
“Arena Training is a onestop shop that records and provides a historical record for each employee. I like that this tool allows quizzes to be added, which holds the employee accountable for actually reading and understanding the material content.”
– Cindy Lalowski, Senior Quality Systems Manager, AEye
Regulated med-tech companies rely on distributed teams of experts to design, manufacture, and support their products. These experts can be direct employees, contractors, or other design and manufacturing partners. Without access to the single product record, the various experts could use different revisions or the wrong documents. Without automated quality and change processes, they could not know about important product updates. The results include extra design cycles, missed release dates, or manufacturing errors.
Arena facilitates teamwork with notifications, dashboards, access control, and a range of collaboration options to share information between all impacted stakeholders. Automated notification can be driven by key actions, such as when ECOs need to be reviewed and approved. Arena also controls access so that internal employees and external partners see only the product information needed to do their specific job.
This access improves innovation and quality while reducing cost and process cycle times. For example, design for manufacturability (DFM) processes are compressed when the manufacturer can review designs earlier and give feedback to source different parts or change the design for ease of manufacturing.
“Arena is used throughout our organization for BOM management and for quality management. We are able to collaborate on solving nonconformance reports and complaints and share BOMs and specifications with our suppliers. Arena also helps us demonstrate RoHS compliance. Every audit by our certification body is supported by Arena’s system, whether using search results for the particular area under question or printouts from reports generated.”
– Avraham Harris, Director Quality Affairs & Regulatory Affairs, Accellix

For many medical device companies, the lack of a well-implemented CAPA process is the primary concern for audit issues, observations, or nonconformance reports (NCRs). CAPA processes are complicated by multiple approval disciplines, difficult root cause closure, and multiple quality processes that require a sophisticated ability to traverse product history for time sensitivities. CAPA processes often trigger an engineering change process to resolve product defects and other issues. A single quality issue may affect one or many products. And one product may have one or many quality issues. Mastering CAPA processes provides significant payback directly tied to company success, but it requires a system that supports the process demands.
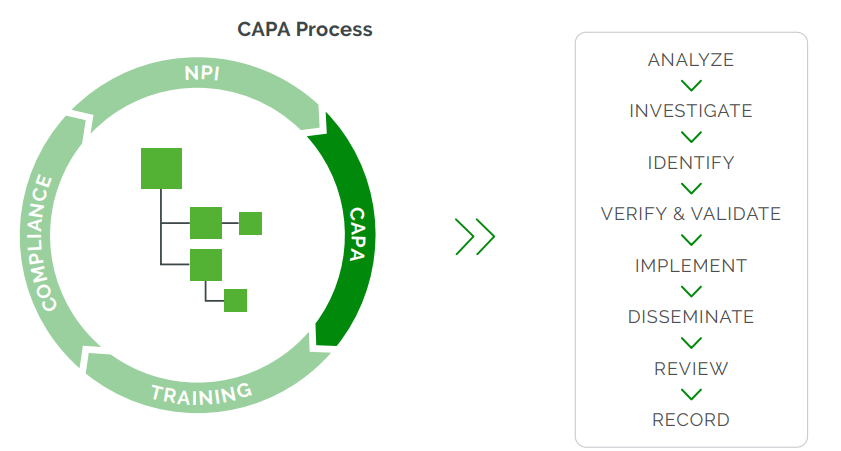
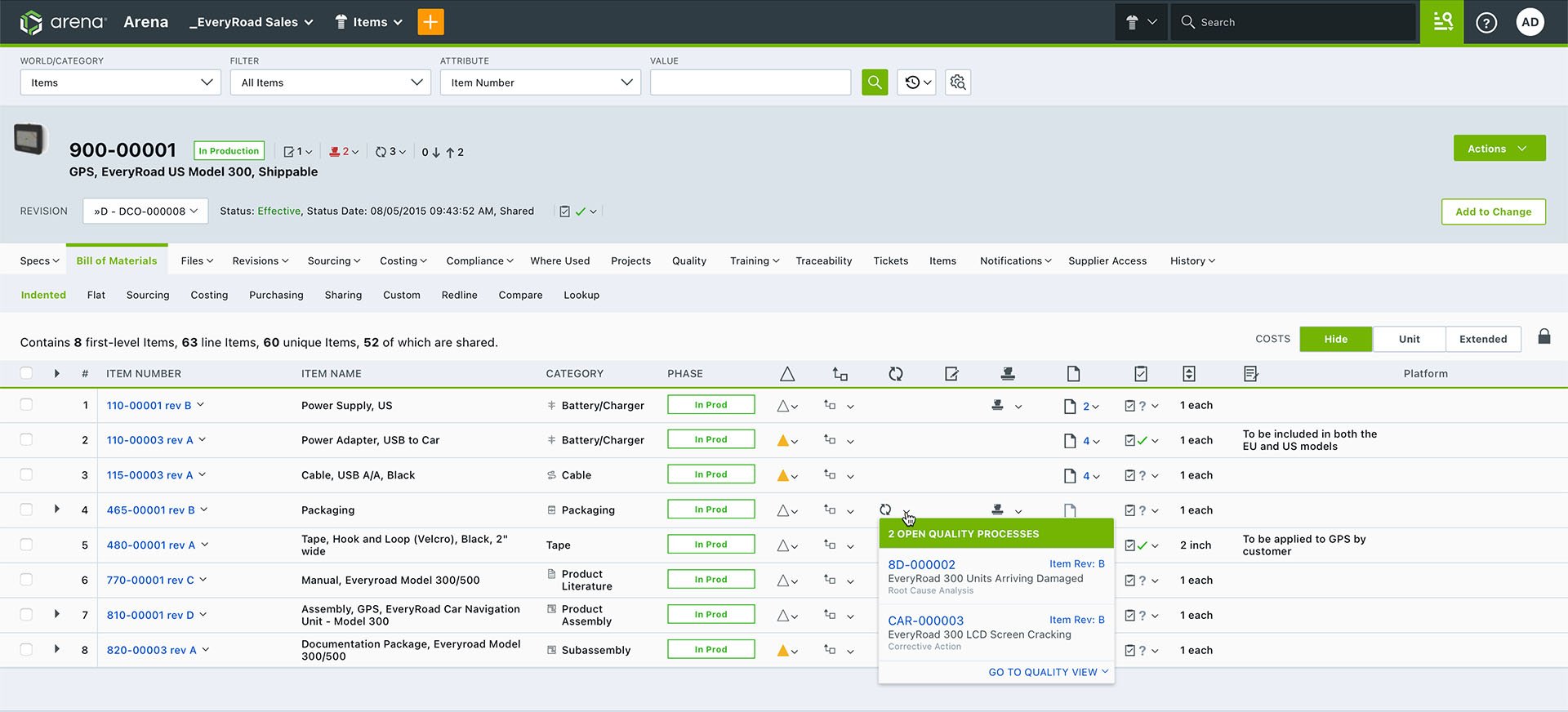
Arena maintains all these relationships via live links, so users can instantly move from documents, parts, and multilevel assemblies to quality issues or vice versa. Arena provides visibility between the CAPA records and related change orders, which may include BOM and AML redlines. In this way, internal auditors, quality and regulatory affairs, and other affected groups can instantly access and view all the issues, actions, and steps through final resolution in one central system.
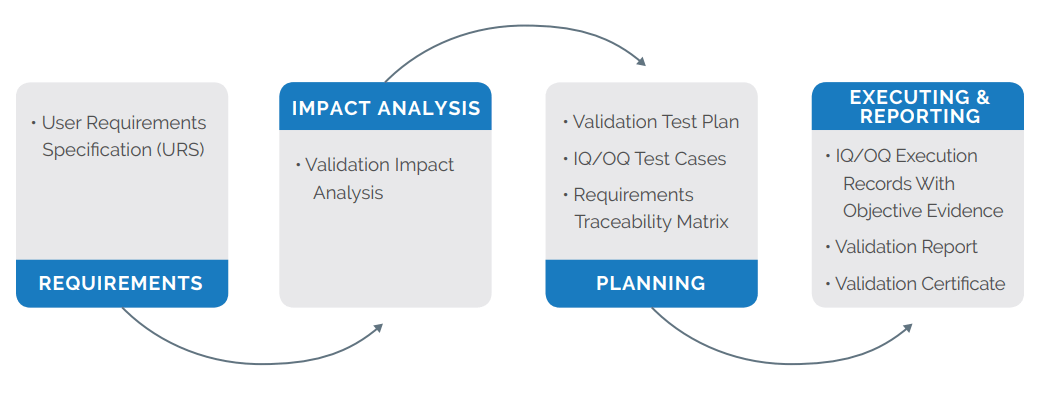
Medical device companies are required to validate enterprise software systems that are part of the quality system for their intended use, according to established protocols (per 21 CFR 820.70(i) and 21 CFR 11.10(a)). Regulated companies understand this requirement means each enterprise system upgrade can have a major impact on business operations. However, enterprise systems must be upgraded or enhanced periodically to meet expanding business requirements, provide technological advantages, and better meet industry demands.
Arena Validate, an industryleading software validation service, is designed to speed FDA 21 CFR Part 820 and Part 11 compliance, which allows your teams to focus on their core competencies and spend significantly less time validating Arena’s software application. We provide the critical elements of change and document management, design controls, electronic records, electronic signatures, and quality management.
“We’ve … added in an impact, a rootcause analysis, and risk management observation process. Each one of these different processes can call the other ones, or demand that another process be used. They can also be linked together so that when you get an audit, you have a tightly integrated cross-linked evidence chain for whatever problem you’re dealing with.”
– Chief Operating Officer, Swan Valley Medical
Arena validates each Arena release against a predefined set of intended uses common to all medical device manufacturers. Arena then distributes the results of validation testing to all subscribers. As a result, Arena’s regulated customers can leverage 100% of the enhancements to yield additional benefits. With Arena’s cloudbased QMS and validation service, you are able to adopt every new software release without the fear of being revision-locked on your QMS software system and without having to complete a difficult upgrade process.
Audits, both internal and external, are a constant reality for medical device product companies. And how well you perform during audits determines not only the success of your team and product but sometimes your company’s very existence. One approach to an audit is to engage valuable people resources in the task. These people may be asked to obsessively and manually collect all of the product and process details and become the historians of the DHF/DMR. Yet, this method exposes you to many failure points (e.g., reliance on tribal knowledge, introduction of human errors, lack of visibility, communication delays). It is also a costly process to maintain as your product lines grow and mature.
“Implementation was made very easy by Arena giving us complete IQ and OQ documents. The wide range of standard out-of-the-box functionality narrowed the scope of our work even more. This resulted in a very productive and complete PQ while requiring much less management than other IT system implementations I have been a part of.”
– Ed Reith, Supply Chain Engineering Manager, Ebb Therapeutics
Arena provides the functionality to achieve and maintain regulatory compliance easily while performing product and quality processes. Peace of mind is a priceless benefit when you have all of your quality actions and change processes linked to the product record. Having the full product design history and product record controlled and tracked in one system provides you confidence during stressful audit processes.